|
The fusion process has been studied as a means of understanding nuclear matter, to learn about the nuclear physics of stellar objects, and to create thermonuclear weapons. For example, on Earth, temperatures in excess of 100 million degrees Celsius are needed to cause fusion between deuterium and tritium. The massive gravitational pull of stars help induce fusion, but without this force, higher temperatures are required to cause fusion. So far, we have looked at the fusion that occurs in stars, but scientists and engineers have been trying to recreate the conditions for fusion here on Earth. Hydrogen, for example, consists of a single proton, whereas the heavy isotopes of hydrogen - deuterium (D) and tritium (T) – release more energy as they contain more parts (Deuterium has one proton and one neutron, while tritium has one proton and two neutrons). With the right amount of heat, a close proximity and high levels of pressure, protons and neutrons can be forced together, releasing different levels of energy depending on their make-up of the element. It is the binding energy between nuclear matter that measures the efficiency with which nucleons are bound together as well as determining the energy released by both fission and fusion. Humans have already used the inverse process, nuclear fission, where heavier elements like uranium and plutonium are split, to create energy. 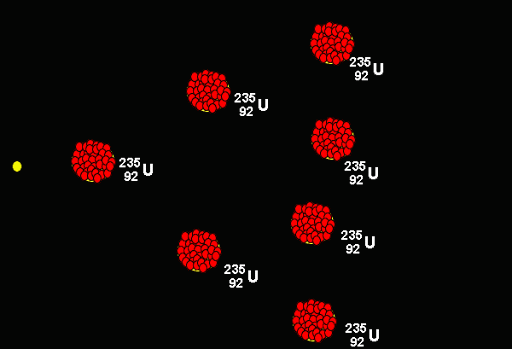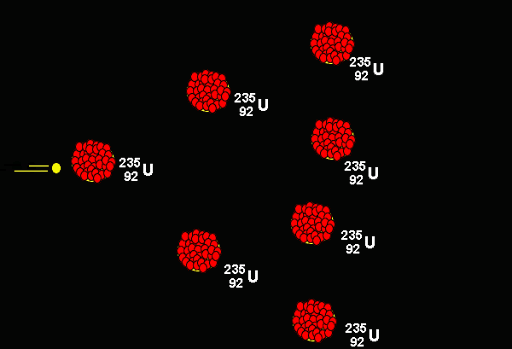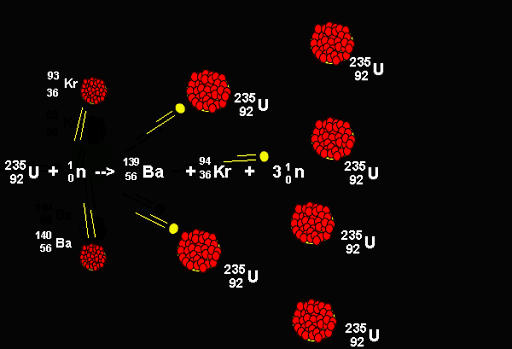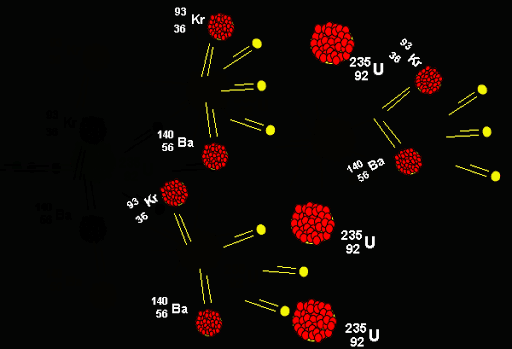 
Research has investigated harnessing this natural reaction and using the energy that is created for our own needs. For each two hydrogen nuclei that are fused to form helium, 0.645% of the mass is carried away as alpha particle kinetic energy and other forms of energy, such as electromagnetic radiation. The core of the Sun fuses 620 million metric tons of hydrogen each second, making 616 million metric tons of helium per second as a result. Stars create energy by fusing hydrogen nuclei and releasing helium. This is not the same for heavier nuclei, which have a shorter-ranged nuclear force, which, instead of releasing energy with fusion, require energy as an input. When protons and neutrons are combined from lighter nuclei by this nuclear attraction, the nuclear reaction releases extra energy. The extreme pressures created by the gravity of stars create this final requirement for fusion to occur. As well as the heat and close proximity, fusion requires the nuclei to be confined in a small area. These high temperatures provide enough energy for light nuclei to overcome their natural electrical repulsion so that, once they come into very close range of each other, the nuclear force that attracts them outweighs the electrical repulsion (known as the Coulomb force), allowing them to fuse. This all creates the perfect conditions, including the high temperatures of around ten million degrees Celsius, required for hydrogen fusion. Stars, including our sun, are balls of plasma made of hydrogen and helium, created by the gravitational collapse of clouds of cold gas, which is compressed and heated, becoming plasma. 
Plasma is made of free moving electrons and positive ions, and has unique properties that are distinct from other gases, liquids, or solids. 
National Structural Integrity Research Centreįusion reactions happen in a hot gas known as plasma.Structural Integrity Research Foundation.The U-236 nucleus then rapidly breaks apart into two smaller nuclei (in this case, Ba-141 and Kr-92) along with several neutrons (usually two or three), and releases a very large amount of energy.Īmong the products of Meitner, Hahn, and Strassman’s fission reaction were barium, krypton, lanthanum, and cerium, all of which have nuclei that are more stable than uranium-235. \): When a slow neutron hits a fissionable U-235 nucleus, it is absorbed and forms an unstable U-236 nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
